Multifunctional chiral silanol ligands for enantioselective catalysis
Abstract
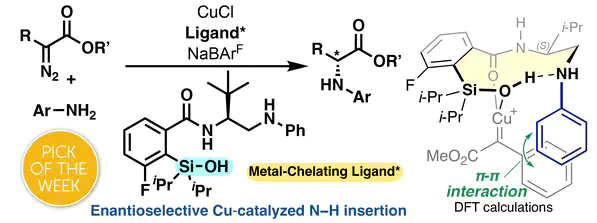
We report transition metal catalysis using novel chiral metal-chelating ligands featuring a silanol coordinating group and peptide-like aminoamide scaffold. The catalytic properties of the silanol ligand are demonstrated through an enantioselective Cu-catalyzed N–H insertion affording unnatural amino acid derivatives in high selectivity. Our investigations into the silanol coordination mode include DFT calculations, ligand structure investigations, and X-ray structure analyses, which support the formation of an H-bond stabilized silanol-chelating copper carbenoid complex. A π–π stacking interaction revealed by DFT calculations is proposed to enable selectivity for aryl diazoacetate substrates, overcoming some of the traditional limitations of using these substrates.