Equivalent Atrop- and Positional Isomerism in Styrene Derivatives Prepared by Enantioselective 1,3-Diarylation
Abstract
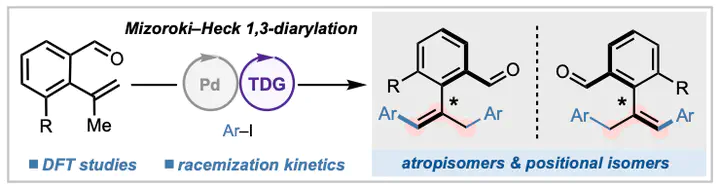
Isomerism, the ability of a single set of atoms within a molecule to exist in different three-dimensional spatial arrangements connecting through distinct bonding networks, gives rise to distinct physical, chemical, and biological properties from a common set of atomic building blocks. While different forms of isomerism are now well appreciated, a rare phenomenon is the coexistence of multiple equivalent forms of isomerism within a given pair of molecules. Here, we report that a simple combination of palladium and amino acid cocatalysts converts ortho-alkenyl benzaldehydes into substituted styrenes possessing equivalent atrop- and positional isomerism. Mechanistically, the reaction proceeds through successive Mizoroki–Heck arylation promoted by the amino acid cocatalyst. DFT calculations show that the atroposelectivity arises from stereoselective β-H elimination in the second arylation cycle, whereas the formation of Z products with constrained rotation about the chiral axis is driven by steric effects at the product stage.