Directed Halogen Atom Transfer (DIXAT): A Powerful Tool for Chemoselective Generation of Aryl Radicals Toward Remote C(sp<sup>3</sup>)–H Functionalization of Aliphatic Amines
Abstract
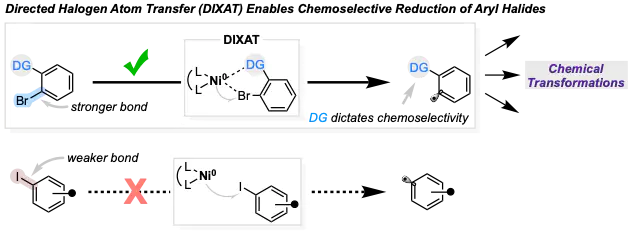
Halogen atom transfer (XAT) and single electron transfer (SET) have emerged as versatile tools for the generation of aryl radicals. The intrinsic reactivity of these methods is governed by bond dissociation energies (BDEs) and reduction potentials, respectively, which typically favor activation of an aryl iodide over an aryl bromide. To date, no existing methods have overturned these inherent reactivity trends. The newly developed strategy, a directed halogen atom transfer (DIXAT), overcomes the above reactivity trends by enabling a chemoselective activation of aryl bromides bearing an ortho directing group, even in the presence of otherwise more reactive aryl iodides. This approach allows for precise control over aryl radical generation based on spatial orientation rather than redox or bond strength considerations. We have harnessed this novel technique to conduct nickel-catalyzed arylation and cascade carboarylation of the rremote C(sp3)–H bonds of aliphatic amines.