De Novo Design, Directed Evolution and Computational Study of Heme-Binding Helical Bundle Protein Catalysts for Biocatalytic Enantioselective Ge–H Insertion
Abstract
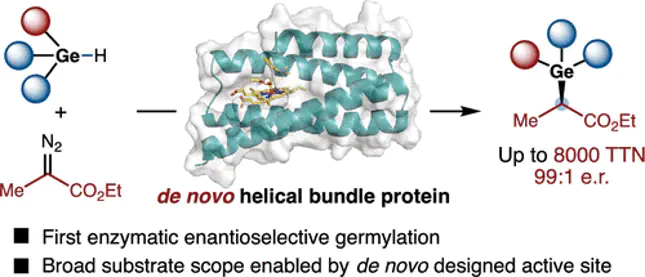
De novo designed proteins offer a malleable platform for the development of stereoselective transformations not found in biochemistry. Here, we report the de novo design and directed evolution of helical bundle protein catalysts for enantioselective germylation through Ge–H insertion, a transformation not previously achieved by enzymatic catalysis. Comparative computational analysis revealed that, relative to Si–H insertion, the Ge–H insertion reaction proceeds through an earlier and more flexible transition state, introducing distinct challenges for stereocontrol. Using a fully de novo designed truncated four-helix bundle scaffold as the starting point, directed evolution afforded a quadruple mutant that catalyzes Ge–H insertion with high efficiency, enantioselectivity, and broad substrate scope. Molecular dynamics simulations indicated that beneficial mutations introduced from directed evolution enhanced active-site preorganization and modulated local backbone flexibility, contributing to improved catalytic performance.