Hexafluoroisopropanol Solvent Effects on Enantioselectivity of Dirhodium Tetracarboxylate-Catalyzed Cyclopropanation
Abstract
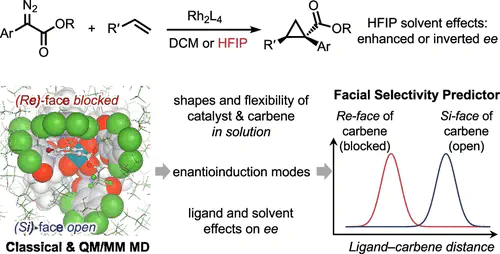
In recent years, additives that modulate both reactivity and selectivity in rhodium-catalyzed reactions of aryldiazoacetates have become increasingly prominent. 1,1,1,3,3,3-Hexafluoroisopropanol (HFIP) has been shown to have a profound effect on rhodium carbene reactivity and selectivity, especially on enabling carbene cyclopropanation in the presence of various nucleophilic poisons. HFIP also has a variable influence on the enantioselectivity of the reactions catalyzed by chiral dirhodium tetracarboxylates, and this study examines the fundamental properties of the rhodium carbene/HFIP system through experimentation, density functional theory (DFT), and molecular dynamics (MD) simulations. These studies revealed that the C4-symmetric bowl-shaped catalysts, which have been previously considered to be relatively rigid, experience far greater flexibility in this hydrogen bonding media, resulting in distortion of the bowl-shaped catalysts. These studies explain why even though a majority of the catalysts have a drop in enantioselectivity in HFIP, some catalysts, such as Rh2(TCPTAD)4, lead to a switch in enantioselectivity, whereas others, such as Rh2(NTTL)4, lead to a considerably enhanced enantioselectivity.