Diversity-oriented photobiocatalytic synthesis via stereoselective three-component radical coupling
Abstract
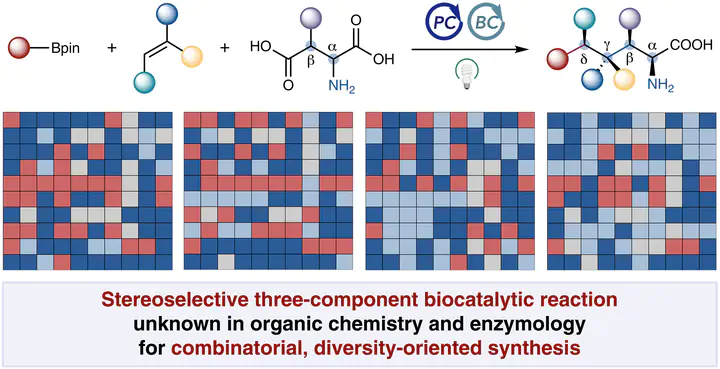
Enzymatic multicomponent C–C bond forming reactions for diversity-oriented synthesis remain rare. Using cooperative photobiocatalysis, we developed a stereoselective three-component radical-mediated C–C coupling unknown in both organic chemistry and biochemistry. Directed evolution of repurposed pyridoxal decarboxylases enabled full fragment variability in this three-component coupling, giving rise to six classes of valuable products, many of which were inaccessible by other methods, even in a racemic fashion. This enzymatic platform integrates a range of asymmetric catalysis principles, including remote stereocenter construction, stereodivergent catalysis, kinetic resolution and parallel kinetic resolution, achieving excellent diastereo- and enantiocontrol over radical intermediates. The broad substrate scope and complementary specificities of evolved enzyme variants enabled combinatorial library synthesis, affording structurally and stereochemically diverse scaffolds for medicinal chemistry.